hM4D(Gi)-mCherry and hM3D(Gq)-mCherry were used for chemogenetics manipulation to inhibit or excite of the GluS1BF neuronal activity. (All viruses were packaged by
BrainVTA)
The viruses used in this article from BrainVTA are in the table below
|
Chemogenetics |
PT-0043 rAAV-Ef1α-DIO-hM4D(Gi)-mCherry-WPRE-pA
PT-0042 rAAV-Ef1α-DIO-hM3D(Gq)-mCherry-WPRE-pA |
Yuping Wang, Peng Cao, Lisheng Mei, Weiwei Yin, Yu Mao, Chaoshi Niu, Zhi Zhang, and Wenjuan Tao
Pub Date: 2019-08-21,
DOI: 10.1016/j.neuroscience.2019.05.034,
Email: [email protected]
Abstract—Trigeminal neuropathic pain (TGN) is an attacking, abrupt, electric-shock headache involving abnormal cortical activity. The neural mechanism underlying TGN remains elusive. In this study, we explored the role of microglia in the primary somatosensory barrel cortex (S1BF), which is a critical region for TGN, of a mouse model of TGN that displayed significant pain-related behaviors. Using electrophysiological recordings, we found robust neuronal hyperactivity in glutamatergic neurons of S1BF (Glu
S1BF). Chemogenetic inhibition of Glu
S1BF neurons significantly relieved mechanical allodynia in TGN mice. In naïve mice, chemogenetic activation of Glu
S1BF neurons induced pain sensitization. In addition, we found that microglia in the S1BF (microglia
S1BF) were significantly activated, with density and morphology changes. Intraperitoneal administration of minocycline, a microglia inhibitor, attenuated pain sensitization, and decreased Glu
S1BF neuronal activity. Together, these findings demonstrate the putative importance of microglia as a key regulator in TGN through actions on Glu
S1BF neuronal adaptation.
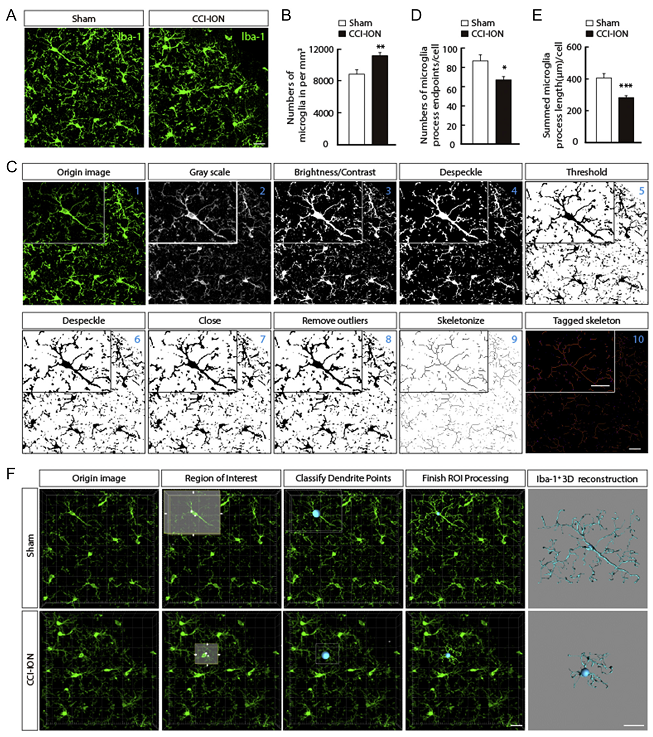
Fig. 1 Activation of microgliaS1BF after CCI-ION.
In this study, using a model of TGN in mice with chronic constriction injury of the infraorbital nerve (CCI-ION), the authors identified changes in the activity of Glu
S1BF and microglia
S1BF cells, and their roles in the development of TGN. Furthermore, combining microglial cell morphological reconstruction, chemogenetic (From
BrainVTA), electrophysiological and pharmacological methods, they described the putative importance of microglia as a key regulator in TGN through actions on GluS1BF neuronal adaptation.
BrainVTA offers viral vector construction & virus packaging services for AAV, LV, RABV, PRV, HSV and VSV that help researchers explore questions about genes, neurons, circuitry structure, function of brain network, mechanism and treatment of diseases.
If you have any needs, just email us at
[email protected].