ChR2 and eNpHR viruses were used for optogenetic manipulation to verify the role of VTA GABAergic neurons in mediating anesthesia. hM3Dq and hM4Di viruses were used for chemogenetics manipulation to further investigate the effect of VTA GABAergic neurons on anesthesia. (All viruses were packaged by
BrainVTA)
The viruses used in this article from BrainVTA are in the table below
|
Optogenetic |
PT-0002 rAAV-Ef1a-DIO-ChR2-mCherry
PT-0007 rAAV-Ef1a-DIO-eNpHR-mCherry |
|
Chemogenetics |
PT-0042 AAV-Ef1a-DIO-hM3Dq-mCherry
PT-0043 AAV-Ef1a DIO-hM4Di-mCherry |
|
Control |
PT-0013 rAAV-Ef1a-DIO-mCherry |
Pub Date: 2019-09-19,
DOI: 10.3389/fncir.2019.00073 Email: [email protected]
Lu Yin, Long Li, Jiao Deng, Dan Wang, YongXin Guo, XinXin Zhang, HuiMing Li, ShiYi Zhao, HaiXing Zhong and HaiLong Dong
The ventral tegmental area (VTA) reportedly regulates sleep and wakefulness through communication with the lateral hypothalamus (LH). It has also been suggested that adequate anesthesia produced by administration of chloral hydrate, ketamine, or halothane significantly reduces the GABAergic neuronal firing rate within the VTA. However, the exact effects on GABAergic neurons in the VTA and the mechanisms through which these neurons modulate anesthesia through associated neural circuits is still unclear. Here, we used optogenetic and chemogenetic methods to specifically activate or inhibit GABAergic neuronal perikarya in the VTA or their projections to the LH in Vgat-Cre mice. Electroencephalogram (EEG) spectral analyses and burst suppression ratio (BSR) calculations were conducted following administration of 0.8 or 1.0% isoflurane, respectively; and loss of righting reflex (LORR), recovery of righting reflex (RORR), and anesthesia sensitivity were assessed under 1.4% isoflurane anesthesia. The results showed that activation of GABAergic neurons in the VTA increased delta wave power from 40.0 to 46.4% (P = 0.006) and decreased gamma wave power from 15.2 to 11.5% (P = 0.017) during anesthesia maintenance. BSR was increased from 51.8 to 68.3% (P = 0.017). Induction time (LORR) was reduced from 333 to 290 s (P = 0.019), whereas arousal time (RORR) was prolonged from 498 to 661 s (P = 0.007). Conversely, inhibition of VTA GABAergic neurons led to opposite effects. In contrast, optical activation of VTA–LH GABAergic projection neurons increased power of slow delta waves from 44.2 to 48.8% (P = 0.014) and decreased that of gamma oscillations from 10.2 to 8.0%. BSR was increased from 39.9 to 60.2% (P = 0.0002). LORR was reduced from 330 to 232 s (P = 0.002), and RORR increased from 396 to 565 s (P = 0.007). Optical inhibition of the projection neurons caused opposite effects in terms of both the EEG spectrum and the BSR, except that inhibition of this projection did not accelerate arousal time. These results indicate that VTA GABAergic neurons could facilitate the anesthetic effects of isoflurane during induction and maintenance while postponing anesthetic recovery, at least partially, through modulation of their projections to the LH.
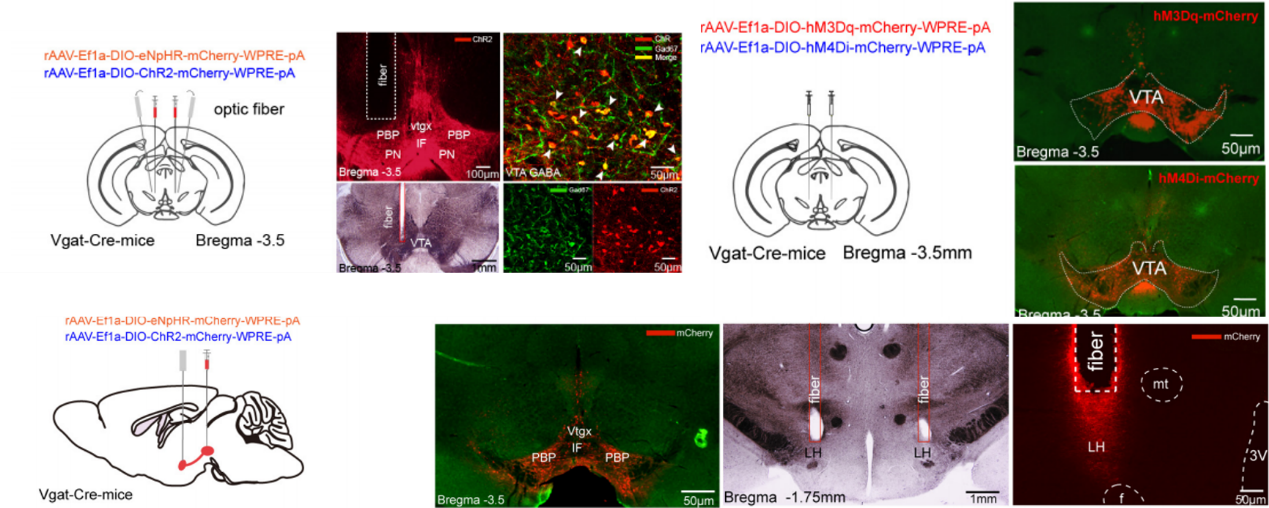 Fig.1 Appling Optogenetic/Chemogenetic manipulation to study the mechanisms of GABAergic neurons in the VTA on general anesthesia
Fig.1 Appling Optogenetic/Chemogenetic manipulation to study the mechanisms of GABAergic neurons in the VTA on general anesthesia
To investigate the role of VTA GABAergic neurons and their projections to the LH in mediating the induction, maintenance, and arousal stages of general anesthesia, the researchers applied optogenetic and chemogenetic strategies (From
BrainVTA), combined with electroencephalogram (EEG) analysis and behavioral tests to study. The results showed that VTA GABAergic neurons and their projections to the LH region are involved throughout the induction, maintenance, and emergence from isoflurane anesthesia.
BrainVTA offers viral vector construction & virus packaging services for AAV, LV, RABV, PRV, HSV and VSV that help researchers explore questions about genes, neurons, circuitry structure, function of brain network, mechanism and treatment of diseases.
If you have any needs, just email us at
[email protected].
